What are the major and minor resonance contributor(s) for the azide anion, #N_3^–#?
1 Answer
The steps for drawing the Lewis structures are:
1. Write all possible connections for the nitrogen atoms.
N-N-N
2. Calculate
3. Calculate
where
4. Draw new structures.
This time insert the double and triple bonds in all possible combinations.
5. Add valence electrons to give each atom an octet.
6. Calculate the formal charge on each atom.
We end up with 3 possibilities.
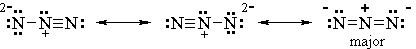
7. Identify the major contributor.
All three structures have separation of charge.
The first two structures have a charge of 2- on one N atom. The third structure has a charge of -1 on two separate N atoms.
The most stable contributor will have the greatest delocalization or spreading out of charge.
So the first two structures are minor contributors, and the third structure is the major contributor.

