What trend in electronegativity do you see as you go across a period/row on the periodic table?
1 Answer
See Below for Answer.
Explanation:
Before I go on, I want to bring up that it's not just electronegativity that is affected as you go across a period or up and down a group. Ionization energy and atomic radius also fluctuate.
Here's a diagram:
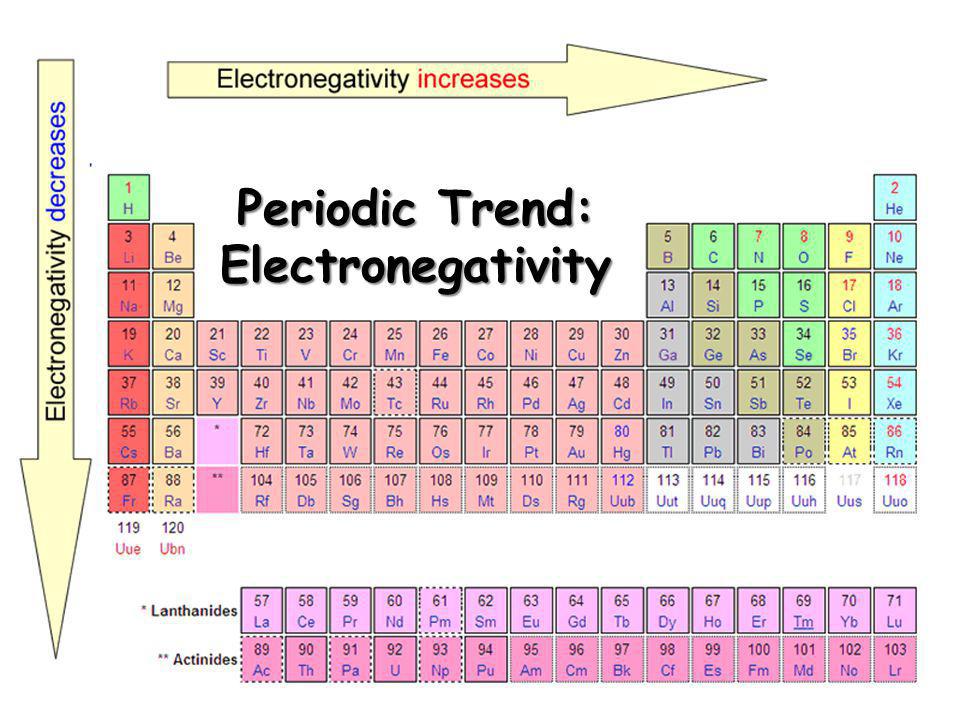
As you can see, electronegativity increases as you go across a period, while electronegativity decreases as you go down a group. However, if this is true (which it is), we can also say many other factors occur, such as:
-
As you go across a period from left to right, electronegativity increases, ionization energy increases, and atomic radius decreases. In order for energy to increase, radius must decrease.
-
As you go up and down a period, electronegativity decreases, ionization energy decreases, and atomic radius increases. In order for energy to decrease, radius must increase.
-
Electronegativity is the force/energy required to acquire electrons and form negative ions during chemical reactions. (definition credit: from my computers dictionary).
-
Ionization Energy is the complete opposite. Ionization is the force/energy required to remove electrons from an ion. These strive to make cations while electronegative ions usually involve anions (hence the phrase electronegativity).

